|
His experiments to determine the nature of positively charged particles, with Francis William Aston, were the first use of mass spectrometry and led to the development of the mass spectrograph. Thomson is also credited with finding the first evidence for isotopes of a stable (non-radioactive) element in 1913, as part of his exploration into the composition of canal rays (positive ions). In 1897, Thomson showed that cathode rays were composed of previously unknown negatively charged particles (now called electrons), which he calculated must have bodies much smaller than atoms and a very large charge-to-mass ratio. Sir Joseph John Thomson OM FRS (18 December 1856 – 30 August 1940) was a British physicist and Nobel Laureate in Physics, credited with the discovery of the electron, the first subatomic particle to be found. This showed that cathode rays consist of negatively charged particles.Owens College (now the University of Manchester) He observed that cathode rays were deflected towards the positive plate of the electric field. Thomson applied an electric field in the path of cathode rays in the discharge tube. DISCOVERY OF ELECTRONS – THOMSON EXPERIMENT AND RESULT He discovered that cathode rays consist of negatively charged subatomic particles (now called electrons), present in all atoms of the elements. J Thomson discovered electrons while studying characteristics of cathode rays. Although, he could not establish what electric current or cathode rays comprise and what is moving from the negative terminal to the positive terminal.įinally, in 1897 J. William observed that when he applies a high electric potential across the discharge tube, then current flows from the negative terminal to the positive terminal in the form of rays. William Crooke, the British Scientist, in the year 1875 successfully created a near to perfect vacuum (0.01 mm of mercury) in a glass tube sealed at both ends with metal plates. William Crook is the first person to pass an electric current through a vacuum 
However, most of them failed as none of them were able to create a perfect vacuum. After carrying out a series of experiments with – solids, liquids, and gases, scientists took one step further to drive electricity through a vacuum. They proved that the electricity or the electric current can flow through any substance – solid, liquid or gas if enough driving force or electric potential exists. The magnitude of the electric current flowing through a substance is directly proportional to the electric potential applied across it. The electric potential is a driving force that results in the flow of current through a substance due to the difference in concentration of charges at two ends of it. Scientists in the early nineteenth century were aware of electricity and the effect of electric potential. Who discovered electrons CATHODE RAY EXPERIMENT To know about the basic structure of an atom, click here 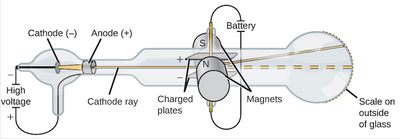
This led to the discovery of the nucleus and other subatomic particles – protons and neutrons. Thomson discovered electrons, the race began among other scientists to uncover the basic structure of an atom. 
In his quest to study properties of cathode rays, he discovered that atoms contain negatively charged subatomic particles – ‘electrons’. 
But things changed after the discovery made by an English scientist named J.J. Thomson, the one who discovered electronsįor a long period in history, scientists were of opinion that atoms could not be broken further.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
